|
But unfortunately, many users don’t add marker names and leave default tube names (Tube_001, Tube_002, etc.). If it were up to me, I would make it a requirement that parameters must be labeled with marker names (CD3-FITC, CD19-PE, etc.) and tubes must contain descriptive labels (WT, KO, treated, untreated, FMO, etc.) before users are allowed to record data. This issue is mainly a pet peeve of mine, but could cause some problems in data analysis. (Image credit: Mario Roederer via The Daily Dongle) 4. Isotype controls identify problems with background staining, but don’t account for spreading error from other fluorophores in the panel. However, once fluorescence minus one (FMO) controls were developed, it was determined that this was a much better method for gating positive cells. Isotype controls were used many years ago to distinguish positive staining from negative staining. For example, ThermoFisher’s AbC beads are more likely to have issues with the polymer dyes (BUV, BV, BB, Super Bright) compared to the UltraComp and UltraComp Plus beads. However, you should check with the manufacturer if your beads have any known fluorophore issues. Unfortunately I have not found a reliable way to predict which fluorophores have an altered emission spectra on beads. Using only compensation beads is not a guarantee that there will be compensation errors, but it is a possibility. In the image below I have a few plots to demonstrate how the single stained beads for this panel are perfectly compensated but the single stained cells are not. The image above shows the consequences of analyzing data that has unmixing errors because only compensation beads were using to calculate compensation – note how the frequencies of the gates differ. If compensation beads are used for all single stained controls, you should be on the lookout for compensation errors in your fully stained cells (see the first bad data blog post for patterns to look for). Because of this mismatch in fluorophore signature, that means a compensation matrix that works well on beads might not work quite as well on cells. Why? No one has any idea and I’m not sure anyone is trying to figure it out, but it is a known fact (it might be written on the technical data sheet of your antibody). For some unknown reason, the emission spectra of a fluorophore is sometimes different if the fluorophore is on a bead vs. However, compensation beads are not a perfect replacement for single stained cells. Only compensation beads run for single stained controls (no single stained cells).Ĭompensation beads are a great tool and can be very useful if a marker is not well expressed on cells or if there aren’t enough cells to use for both samples and controls. It may be possible to get useable data when reusing old controls, but this should be tested before it is put into practice.Ģ. 
But if you’re going to skip the single stain controls you should definitely know that this is not best practice and you should have a very good understanding of the consequences of analyzing data with compensation errors (a future blog post will be dedicated to this topic!). I’ll admit that I don’t run controls 100% of the time. Unfortunately it’s somewhat common for people to run experiments without controls and apply an old compensation matrix to new samples. These variations could mean that the compensation matrix needs to be different, and the only way to properly create or adjust a compensation matrix is with single stain controls. From one experiment to the next, there may be variations in the antibody staining, fluorophore stability, and/or instrument stability. Best practice says single stain controls must be run every single time you run an experiment. 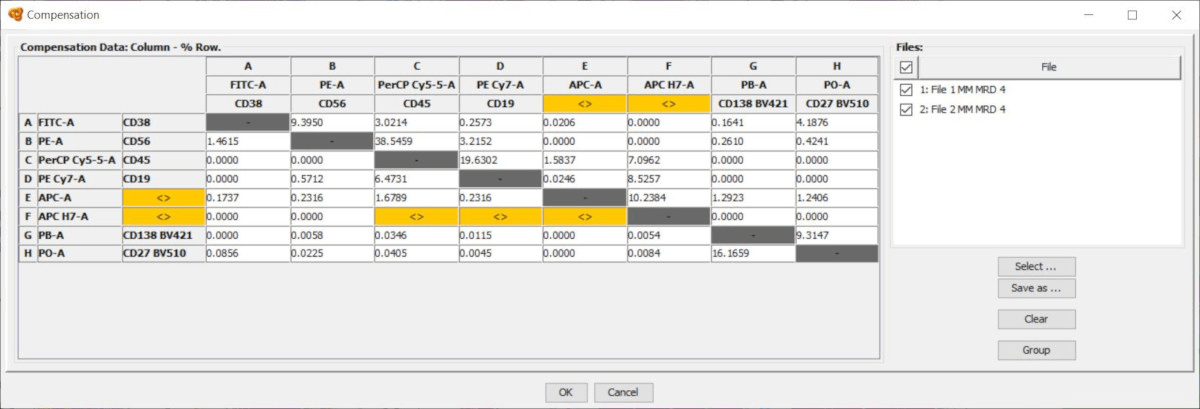
This scenario would occur when someone runs controls and samples on one day, but then repeats the experiment on another day and uses the compensation matrix from the first experiment instead of bringing new controls. The above image shows an example of two samples with the same panel using the same compensation matrix but stained and run on the cytometer on different days.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
